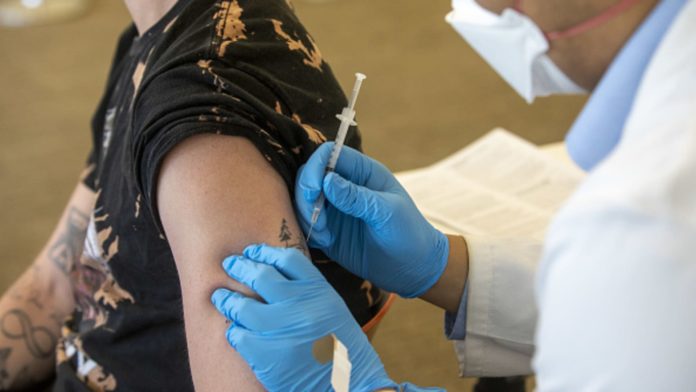
A health worker administers a dose of the Bavarian Nordic A/S Jynneos monkeypox vaccine at a vaccination site in West Hollywood, California, on Wednesday, Aug. 3, 2022.
Jill Connelly | Bloomberg | Getty Images
The Food and Drug Administration on Tuesday expanded its authorization for the monkeypox vaccine in a way that would significantly boost the limited supply of shots.
The FDA will allow health-care providers to administer the shots through intradermal injection, or between the layers of the skin, which will increase the supply of doses by as much as fivefold. This type of injection would use one-fifth the normal dose. The intradermal injections are only for adults.
The emergency authorization also allows people under age 18 to receive the vaccine if they are at high risk of monkeypox infection. People under age 18 would receive the shot through subcutaneous injection, or beneath the…